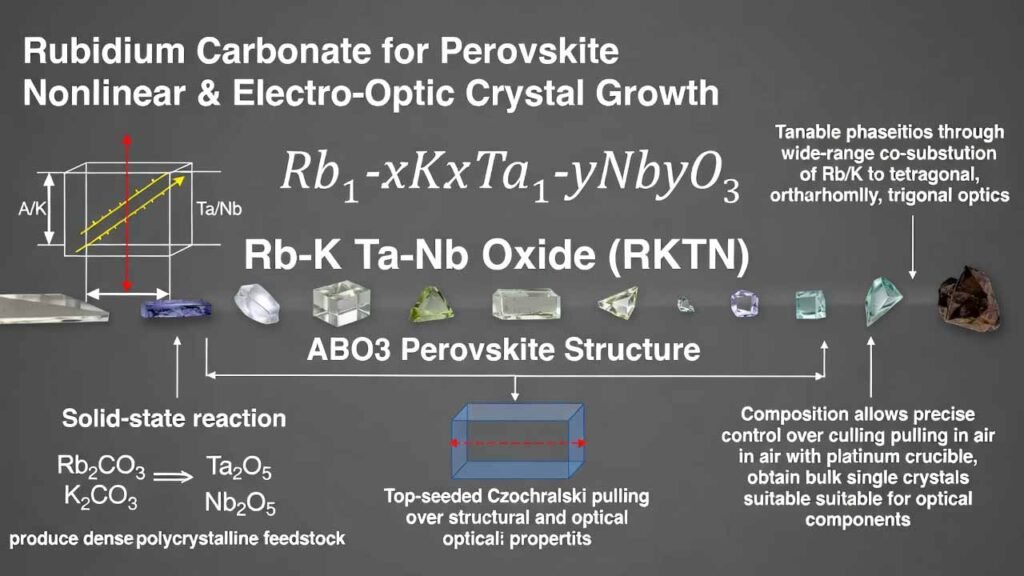
Rubidium Carbonate for Perovskite Nonlinear & Electro-Optic Crystal Growth: Rb–K Ta–Nb Oxide (RKTN, ABO3)
1) Overview
RKTN refers to a family of perovskite-structured oxide single crystals with the general composition Rb1-xKxTa1-yNbyO3 (0 ≤ x < 1, 0 < y < 1). In this ABO3 lattice, Rb/K occupy the A-site and Ta/Nb occupy the B-site, enabling wide-range co-substitution to tune structure and properties. As temperature decreases, typical compositions can pass through multiple phase transitions (cubic → tetragonal → orthorhombic → trigonal), supporting strong dielectric and electro-optic behavior.
A practical manufacturing route combines: (i) solid-state reaction using Rb2CO3, K2CO3, Ta2O5, Nb2O5 to produce dense polycrystalline feedstock, and (ii) top-seeded Czochralski pulling in air (platinum crucible) to obtain bulk single crystals suitable for optical component fabrication.
2) Detailed Experimental Process
A. Raw Materials & Target Stoichiometry
- Rubidium source: high-purity Rb2CO3
- Potassium source: high-purity K2CO3
- B-site oxides: high-purity Ta2O5 and Nb2O5
- Composition design: choose x and y for Rb1-xKxTa1-yNbyO3 based on required phase behavior and electro-optic response.
Practical charge balance guideline: to compensate A-site volatility and improve densification, a slight A-site carbonate excess is used.
| Parameter | Recommended Range | Notes (Process-Relevant) |
|---|---|---|
| (Rb2CO3 + K2CO3) : (Nb2O5 + Ta2O5) molar ratio | 1.1–1.3 : 1 | Adjusted by x: 1.1–1.15 when 0 ≤ x < 0.35; 1.2–1.3 when 0.35 ≤ x < 1 |
| Example weighing ratio (molar) | Rb2CO3 : K2CO3 : Ta2O5 : Nb2O5 = 0.3 : 0.9 : 0.3 : 0.7 | Produces an example composition close to Rb0.1K0.9Ta0.6Nb0.4O3 |
B. Solid-State Synthesis of RKTN Polycrystalline Feedstock (Two-Step Calcination)
- Weighing: weigh Rb2CO3, K2CO3, Ta2O5, Nb2O5 according to the selected x and y, using the carbonate-to-oxide molar ratio guidance above.
- Homogeneous mixing: mix thoroughly (ball milling is commonly applied for uniformity) until the powder blend is fully homogenized.
- Compaction: press the mixed powder into blocks at 30–50 MPa to improve solid-state reaction contact and reduce porosity.
-
First calcination (pre-sintering): place compacts in a platinum crucible and heat in air:
- When 0 ≤ x < 0.35: 750–825°C for 8–12 h
- When 0.35 ≤ x < 1: 850–930°C for 8–12 h
- Example condition: 850°C for 8 h
- Regrinding: cool, remove the calcined body, grind back into fine powder, and remix to eliminate compositional gradients.
- Re-compaction: press again into blocks (30–50 MPa) for the second calcination.
-
Second calcination (densification / phase completion): in a platinum crucible, heat in air:
- When 0 ≤ x < 0.35: 850–900°C for 10–15 h
- When 0.35 ≤ x < 1: 950–975°C for 10–15 h
- Example condition: 975°C for 10 h
- Outcome: obtain RKTN polycrystalline feedstock suitable for melt growth. During reaction, carbonates decompose and release CO2, driving formation of the ABO3 oxide phase.
C. Top-Seeded Czochralski Growth of Bulk RKTN Single Crystal (in Air)
- Crucible & atmosphere setup: load the polycrystalline feedstock into a platinum crucible in a single-crystal pulling furnace; use air (ambient atmosphere).
-
Melting: heat to fully melt the charge:
- General range: 1150–1500°C to melt (composition-dependent)
- Example: melt observed around 1180°C, then raise to about 1250°C and hold
- Superheat hold: maintain a stable high temperature for about 2 hours to homogenize the melt.
- Seeding temperature adjustment: lower to the seeding temperature (example: about 1175°C), then introduce a seed crystal from the top.
-
Growth sequence: perform the standard pulling stages:
- Necking (to eliminate defects)
- Shouldering (to expand to target diameter)
- Constant-diameter growth (stable growth of the bulk crystal)
-
Key growth parameters:
- Pull rate: 0.25–0.5 mm/h (example: 0.3–0.5 mm/h)
- Crystal rotation: 4–10 rpm (example: 5 rpm)
- Constant-diameter temperature window: typically within 1200–1450°C depending on composition (example control: 1270–1280°C)
- Growth duration: 5–10 days (example: 5 days)
- End of growth: separate the crystal from the melt surface, then hold at constant temperature for 1–2 hours to relieve thermal stress.
-
Controlled cooling to room temperature: select cooling schedule based on Curie temperature (Tc):
- If Tc ≤ 25°C: cool at 10–15°C/h down to room temperature
- If Tc > 25°C: near Tc, slow to 1–2°C/h down to room temperature
- Example: cooling at ~1°C/h to room temperature while maintaining rotation
D. Sample Preparation & Property Verification (R&D Workflow)
- Crystal shaping: bulk crystals are typically obtained as rectangular/cubic column forms with {100}-family faces exposed, which simplifies orientation and device cutting.
- Oriented wafer cutting: process into X-plate, Z-plate, and angled cuts (e.g., 15°, 30°, 45°) depending on the electro-optic device design and measurement direction.
- XRD phase confirmation: grind a small portion (fine powder) and compare powder diffraction against expected perovskite ABO3 pattern for phase purity and structural match.
- Dielectric vs temperature (Curie behavior): measure capacitance with an LCR meter to obtain dielectric constant vs temperature (phase-transition mapping).
- Electro-optic test example: using a He–Ne 633 nm laser setup (polarizer → crystal → analyzer → detector), apply an electric field transverse to the growth direction and determine half-wave voltage to calculate the quadratic electro-optic coefficient (reported near room temperature on the order of 10-14 m2/V2 for representative composition).
3) Comparison Summary: This Production Route vs Traditional Approaches
What’s “traditional” in ABO3 ferroelectric crystal development
- Single-site tuning: many established solid solutions adjust only A-site (e.g., A-site substituted KTaO3-based systems) or only B-site (e.g., KTa1-xNbxO3), which limits the design space.
- Less flexible phase engineering: fewer independent knobs to position Curie temperature, phase boundaries, dielectric response, and electro-optic coefficients simultaneously.
- Feedstock preparation can be simpler: sometimes single calcination or narrower composition ranges are used, trading off homogeneity and compositional control.
How the RKTN route differs (and why it matters)
- Dual solid-solution design: simultaneous A-site (Rb/K) and B-site (Ta/Nb) substitution expands the compositional design window for property optimization.
- Two-step calcination: regrinding + second firing improves homogeneity and reaction completeness, which is critical before melt growth.
- Top-seeded pulling for bulk quality: controlled necking/shouldering/constant-diameter growth supports bulk single crystals suitable for optical component fabrication and repeatable electro-optic measurements.
- Cooling schedule tied to Tc: managing phase transitions during cool-down reduces stress/cracking risk and preserves functional domain/phase stability near room temperature.
4) Why Rubidium Carbonate (Rb2CO3) Is Advantageous in This Application
- Direct, clean A-site rubidium supply for ABO3 formation: Rb2CO3 reacts with Ta2O5/Nb2O5 (along with K2CO3) to form the perovskite oxide while releasing CO2, helping drive the solid-state synthesis toward the target phase.
- Stoichiometry control that directly impacts electro-optic performance: in RKTN, Rb content defines A-site occupancy and influences phase transition temperatures and dielectric/electro-optic responses. Using high-purity Rb2CO3 supports precise x-control across Rb1-xKx compositions.
- Compatibility with two-step calcination for high homogeneity: carbonate-based batching pairs well with repeated grind-and-fire cycles, improving cation distribution uniformity prior to melting—an essential prerequisite for stable Czochralski growth.
- Process-friendly in high-temperature oxide crystal production: Rb2CO3 is routinely handled as a solid precursor for oxide synthesis and integrates smoothly into press-and-sinter workflows (30–50 MPa compaction, platinum crucibles, air firing).
- Enables broader property engineering than K-only systems: introducing Rb through Rb2CO3 expands the A-site design space beyond K-based perovskites, supporting more flexible tuning of phase sequences near room temperature where electro-optic devices often operate.
- Supports scalable bulk crystal growth: when the feedstock chemistry is stable and homogeneous, top-seeded pulling at controlled rates (0.25–0.5 mm/h) and rotations (4–10 rpm) can yield bulk single crystals suitable for wafering and device prototyping, such as quadratic electro-optic modulation components.
For R&D teams optimizing RKTN, the rubidium precursor is not a minor detail: the purity, moisture control, and batching accuracy of Rb2CO3 directly influence phase purity of the polycrystalline feedstock, melt stability during pulling, and the repeatability of dielectric/electro-optic measurements. The synthesis method mentioned in this article references patent document number CN202510093878.2
