Rubidium Iodide (RbI) Additive Electrolyte for Aqueous Zinc-Ion Batteries: Dendrite Suppression, Longer Cycling, and Safer Cells
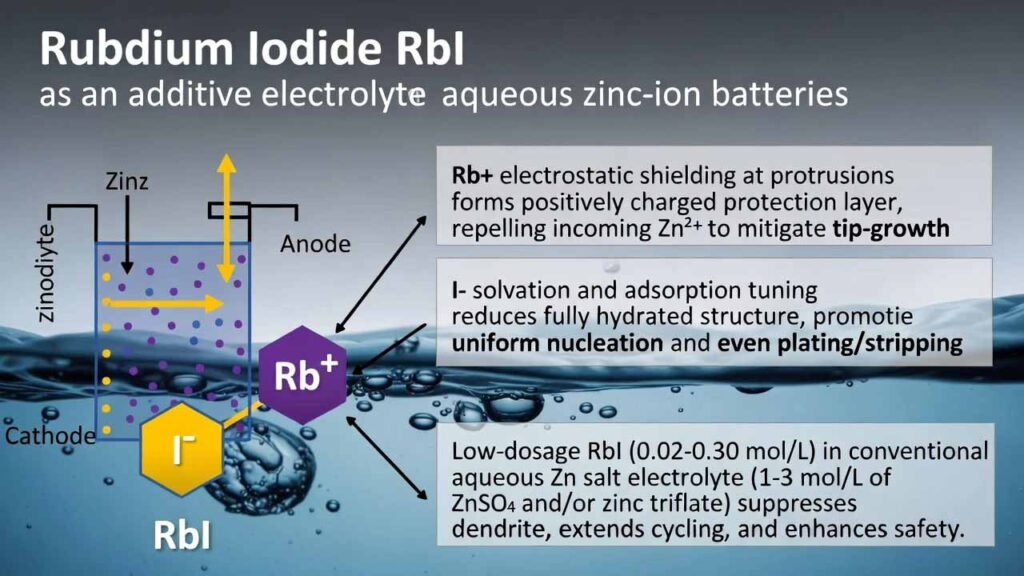
This electrolyte strategy introduces rubidium iodide (RbI) as a low-dosage additive (0.02–0.30 mol/L) into a conventional aqueous Zn salt electrolyte (1–3 mol/L of ZnSO4 and/or zinc triflate). RbI delivers a dual-ion interface regulation effect
Rubidium Iodide (RbI)–Enabled High-Speed Blade Coating for Scalable Perovskite Solar Cells in Humid Air

Perovskite solar cells (PSCs) are attractive for next-generation photovoltaics because they can be made by solution processing, potentially lowering capex and enabling large-area manufacturing. A core scale-up bottleneck is forming uniform, dense perovskite films over larger substrates without sacrificing device performance or stability—especially when moving from small-area lab spin coating to scalable coating methods.
Rubidium Iodide (RbI) Additive Engineering for MA-Free Wide-Bandgap Perovskite Tandem Solar Cells
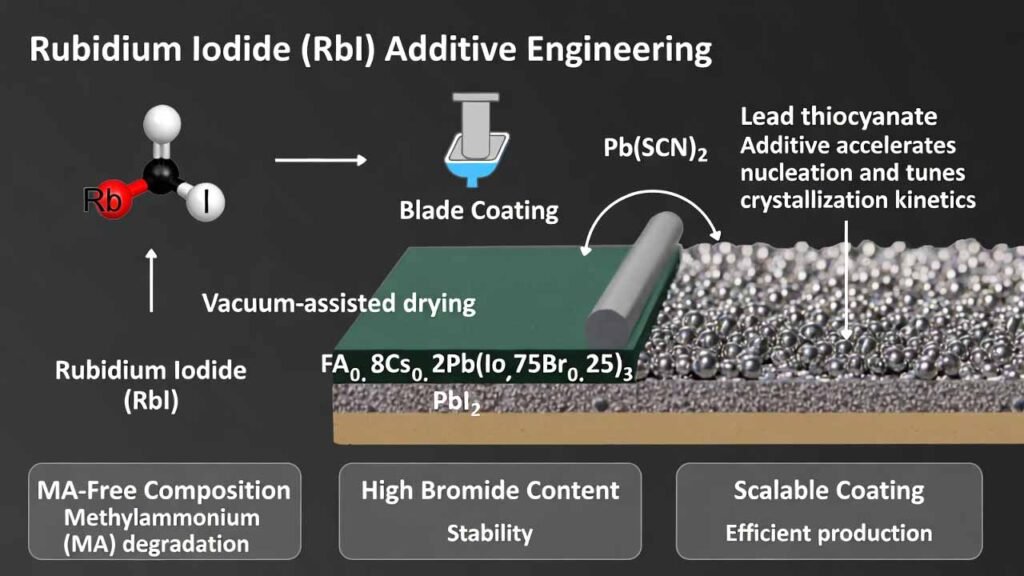
The approach here uses a methylammonium (MA)-free wide-bandgap composition FA0.8Cs0.2Pb(I0.75Br0.25)3 (bandgap ≈ 1.7 eV) to avoid MA-related degradation risk, and applies an additive pair: Pb(SCN)2 plus a rubidium salt (preferably rubidium iodide, RbI). Pb(SCN)2 accelerates nucleation and tunes crystallization kinetics, while the rubidium salt works synergistically to improve grain coalescence, suppress bulk PbI2 residue, and support dense, pinhole-free films suitable for scalable coating.
Rubidium Hydroxide–Enabled Antibacterial Additive for Dark Plastic Pipes (Water Supply & Drainage)

This process builds a dark-pipe-specific antibacterial additive by first using rubidium hydroxide (RbOH) aqueous solution to dissolve lignin and form a rubidium–lignin salt (rubidium ligninate) with highly uniform rubidium distribution at the molecular level. A controlled oxygen-atmosphere thermal treatment then converts the precursor into a brown/dark powder that blends efficiently with carbon black, coupling agents, compatibilizers, and polyolefin resin to produce an extrudable additive. When dosed into pipe formulations, the additive provides antibacterial functionality while also supporting improved composite compatibility and mechanical performance.
Rubidium Hydroxide–Enabled Ternary Copper Sulfide Catalysts for CO2 Electroreduction to Formate
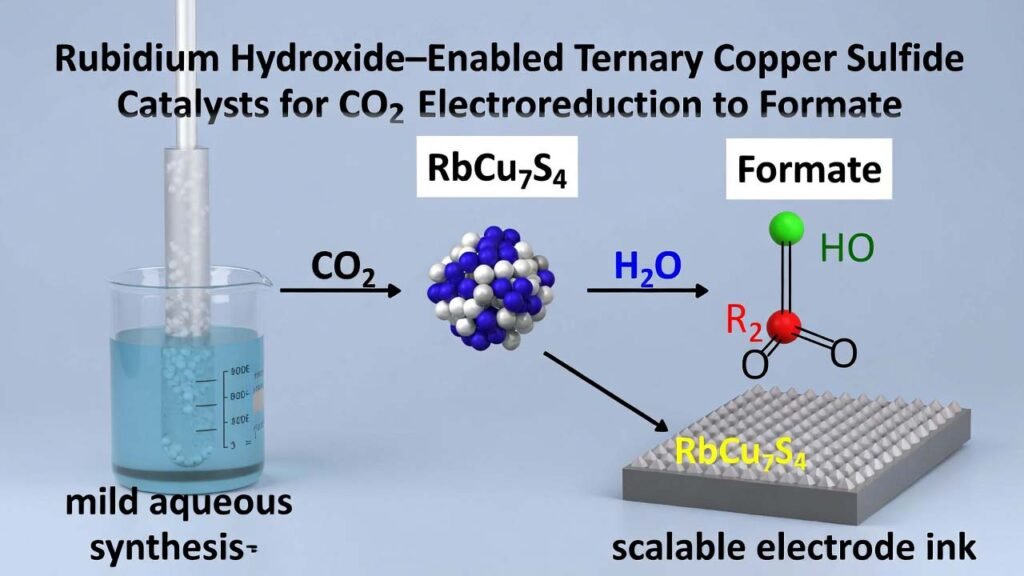
This workflow describes a low-temperature solution synthesis of ternary copper sulfide catalysts (M–Cu–S, M = Na, K, Rb), followed by electrode fabrication and CO2 electroreduction testing. The rubidium-containing phase (RbCu7S4) is produced by using rubidium hydroxide (RbOH) as the alkali metal source in water, enabling incorporation of Rb+ into the catalyst lattice. This helps the catalyst maintain its composition and structure during CO2 electroreduction, while improving formate selectivity and long-term stability compared with conventional copper oxides/sulfides.
Rubidium Hydroxide in Rubidium Metasilicate for GC Nitrogen–Phosphorus Detector (NPD) Performance

This workflow prepares rubidium metasilicate via an aqueous reaction using rubidium hydroxide (RbOH) as the rubidium source, followed by freeze-drying and inert-gas annealing to obtain a stable powder suitable for detector fabrication. The approach emphasizes process control (pH, clarity/transmittance, moisture, annealing atmosphere, and cooling rate) to improve product consistency and yield, and to support longer detector service life during GC operation.
Rubidium Hydroxide (RbOH) in Vanadium Redox Flow Batteries: Stabilizing High-Concentration Negative Electrolytes

A practical way to restore stability is to add a soluble alkaline agent that is compatible with the sulfuric-acid vanadium electrolyte system and does not form precipitates. Rubidium hydroxide (RbOH) is one option in this alkaline set; it supplies strong, fast neutralization via OH− while keeping added species fully soluble in sulfate media (rubidium salts remain soluble under typical VRFB acid ranges).
Rubidium Nitrate in Ultra-Low-Temperature SCR DeNOx Catalysts for Sulfur- and Water-Containing Flue Gas

The catalyst is built from three functional parts: (i) a high-surface-area, hydrophobic and selective carrier (ZSM-5 + TiO2 + modified diatomite, further treated with a silane coupling agent), (ii) an active phase generated from copper/nickel/chromium nitrates synergized with rubidium nitrate through mixing, short combustion, and calcination to create highly dispersed mixed oxide catalytic centers, and (iii) an additive package (cationic surfactant + ammonium heptamolybdate) to enhance anti-poisoning and stabilize performance under complex flue-gas components.
Rubidium Nitrate-Enabled Microwave Hydrothermal Ion Exchange for Ultra-Thin Chemically Strengthened Display Cover Glass

This process performs ion exchange by immersing the glass in an aqueous ion-exchange solution under a microwave hydrothermal environment. The solution is formulated from potassium nitrate, potassium silicate, rubidium nitrate, and cesium nitrate. Microwave hydrothermal conditions accelerate ion transport and diffusion, improving the exchange rate and reducing overall processing time while maintaining good optical transmission.
Rubidium Nitrate–Impregnated Activated Carbon Catalyst for Trifluoroiodomethane (CF3I) Fire Suppression Agent Synthesis

This workflow integrates rubidium nitrate (RbNO3) as the active salt on activated carbon and uses dynamic oxygen dosing to continuously balance two competing needs: (i) oxidizing deposited carbon to keep pores open and active sites exposed, and (ii) preventing excessive oxidation (burn-off) of the activated carbon support.
