Rubidium Nitrate in UV Nonlinear Optics: Step-by-Step Growth of Rb3B6O10NO3 Crystals for Solid-State Laser Harmonic Generation

In this workflow, rubidium nitrate (RbNO3) is a key feedstock because it is simultaneously: (1) the only explicitly defined nitrate (NO3−) source for building the mixed-anion framework, and (2) a rubidium (Rb+) contributor that helps lock stoichiometry and phase formation when paired with boron–oxygen precursors. For R&D and process scale-up, nitrate incorporation control is often the difference between phase-pure powder and mixed phases that limit UV transmission and device yield.
Rubidium Nitrate–Modified Low-Temperature SCR Honeycomb Catalyst for Synergistic NOx Removal & Hg0 Oxidation
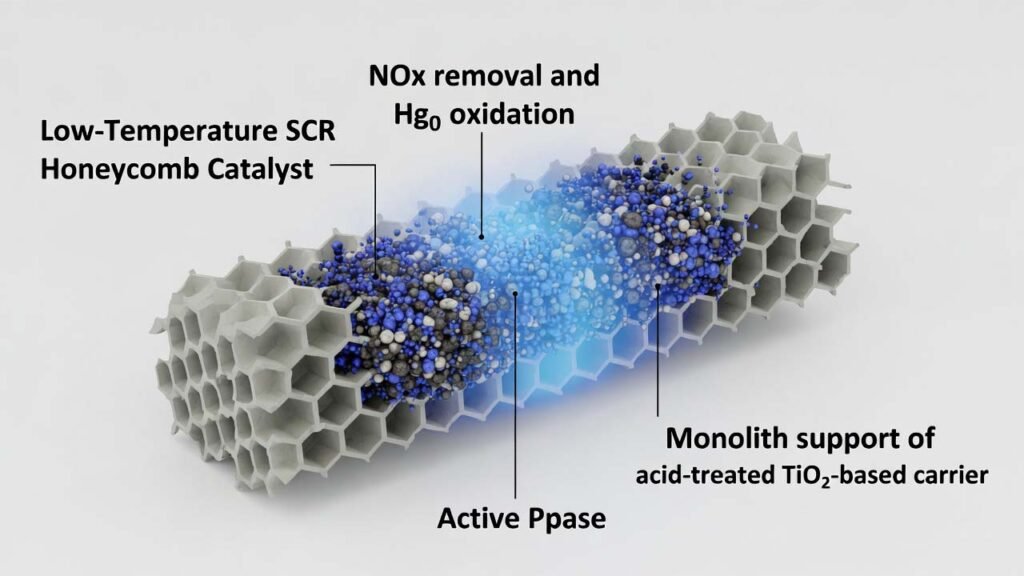
This process prepares a modified honeycomb SCR catalyst composed of a porous monolith support (acid-treated TiO2-based carrier) and a multi-component active phase introduced by impregnation. Rubidium nitrate (RbNO3) is incorporated directly into the carrier formulation to enhance sulfur resistance and stability under low-temperature conditions, while V–W–Mo and Mn/Ce plus trace noble-metal nitrates improve low-temperature SCR activity and Hg0 oxidation performance.
Rubidium Carbonate (Rb₂CO₃) in Catalysis & Materials Science: A Promoter for Microwave-Assisted Carbon-Supported Single-Atom Materials

A practical solution is to combine ZnCl₂ (as a microwave absorber) with a promoter to amplify microwave heating and raise the effective pyrolysis temperature rapidly. In this workflow, Rubidium Carbonate (Rb₂CO₃) can be selected as the promoter to enhance the heating efficiency of the ZnCl₂-assisted system, enabling short, air-atmosphere microwave treatment to produce highly carbonized carbon-supported single-atom materials, followed by simple washing to remove residual salts.
Rubidium Carbonate for Perovskite Nonlinear & Electro-Optic Crystal Growth: Rb–K Ta–Nb Oxide (RKTN, ABO3)
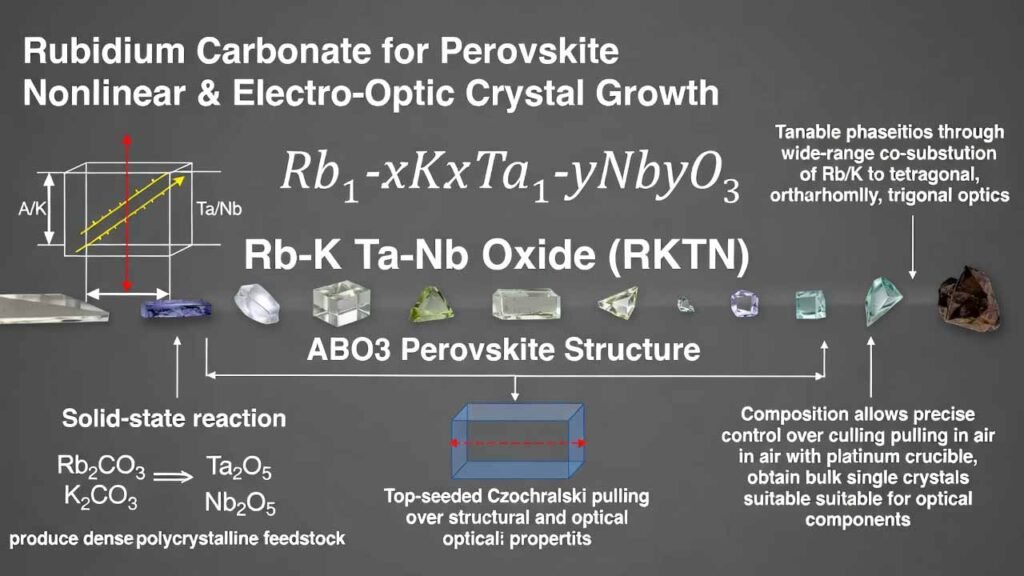
A practical manufacturing route combines: (i) solid-state reaction using Rb2CO3, K2CO3, Ta2O5, Nb2O5 to produce dense polycrystalline feedstock, and (ii) top-seeded Czochralski pulling in air (platinum crucible) to obtain bulk single crystals suitable for optical component fabrication.
Rubidium Carbonate–Enabled Low-Temperature LFP Cathode Materials for Power Batteries and Extreme-Climate Energy Systems
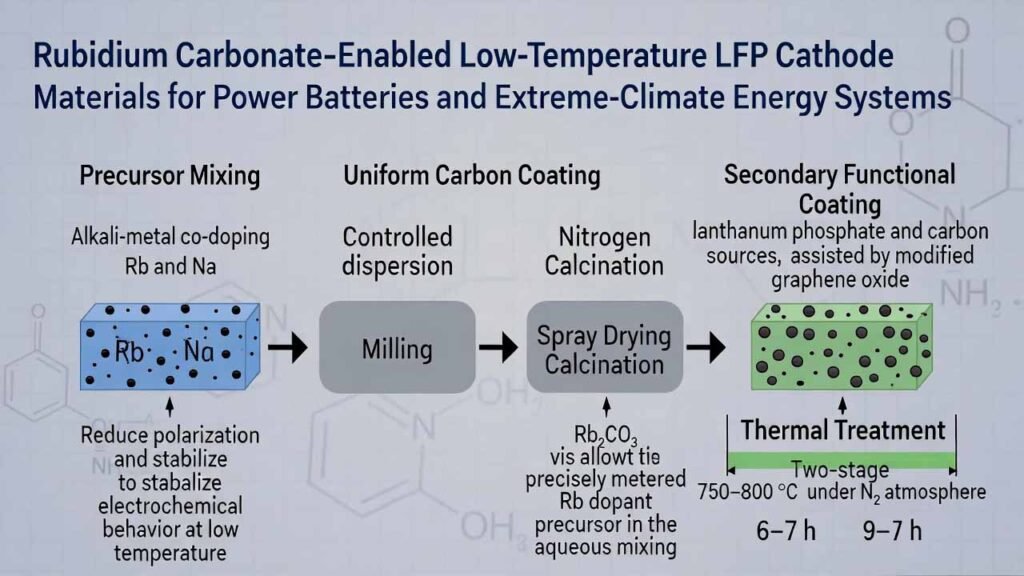
Alkali-metal co-doping (Rb and Na) during precursor mixing to reduce polarization and stabilize electrochemical behavior at low temperature.
Uniform carbon coating formed through controlled dispersion, milling, spray drying, and nitrogen calcination.
Secondary functional coating using lanthanum phosphate and carbon sources, assisted by modified graphene oxide for better compatibility and dispersion.
Rubidium Carbonate (Rb₂CO₃) Bilayer Interfacial Engineering for High-Performance Perovskite Solar Cells
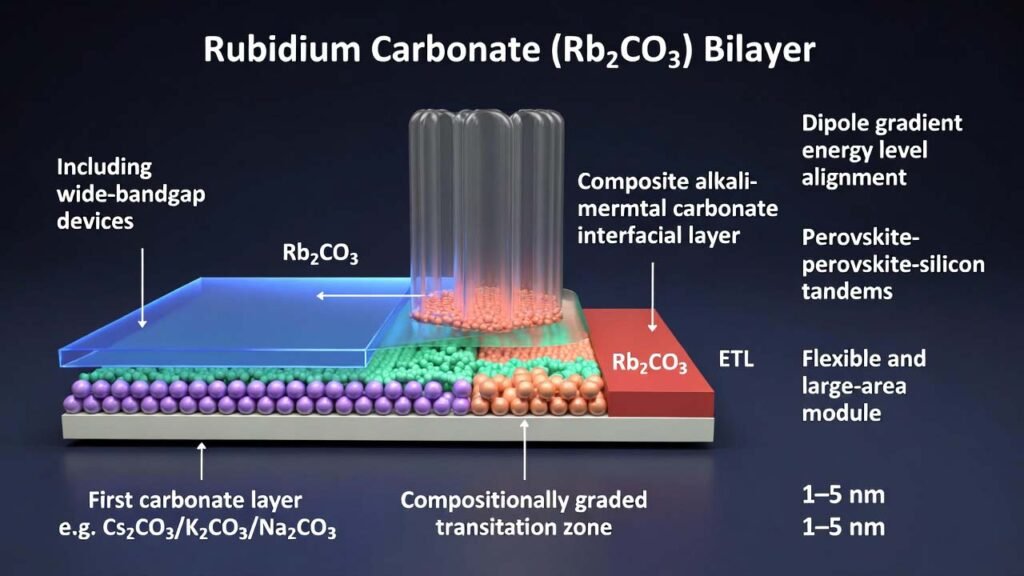
This process builds a bilayer alkali-metal carbonate interfacial stack with a compositionally graded transition zone: a first carbonate layer is placed adjacent to the perovskite (e.g., Cs2CO3/K2CO3/Na2CO3), while a second carbonate layer is placed adjacent to the ETL and can be rubidium carbonate (Rb2CO3). By continuously cross-ramping evaporation rates, the interface dipole changes gradually through thickness, enabling a total interlayer thickness of 1–5 nm while preserving strong charge transport and integrated interface functionality.
Rubidium Chloride (RbCl) for Near-Infrared Luminescent Neodymium Borate Crystals in Photonics

RbCl serves as a highly soluble alkali-metal chloride additive that reshapes the solution/flux micro-environment, promoting polyborate cluster assembly and suppressing failure modes common in low-temperature rare-earth borate attempts (such as rare-earth hydrolysis and precipitation).
Rubidium Chloride (RbCl) in Air-Processed 3D Perovskite Blue-Emissive Thin Films for PeLED Emission Layers
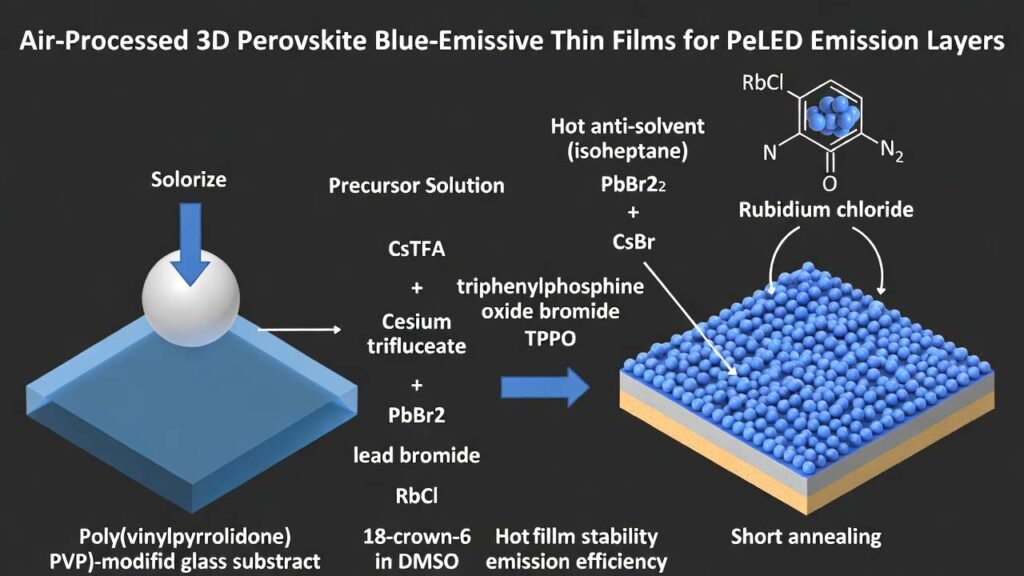
Metal-halide perovskites are attractive emissive-layer materials due to solution processability and tunable bandgap. A key barrier to scale-up is that many high-performance perovskite light-emitting films are prepared in inert environments (e.g., gloveboxes) to avoid moisture-driven defects during crystallization. The workflow below enables 3D perovskite blue-emissive thin films fabricated directly in air by combining: (i) a poly(vinylpyrrolidone) (PVP)-modified glass substrate, (ii) a DMSO-based heated precursor solution containing cesium trifluoroacetate (CsTFA), lead bromide (PbBr₂), rubidium chloride (RbCl), and 18-crown-6 (optionally with triphenylphosphine oxide (TPPO) and cesium bromide (CsBr)), and (iii) a hot anti-solvent (isoheptane) drip during spin-coating plus a short anneal.
Rubidium Chloride–Enabled Uniformly Oriented Quasi-Single-Crystal Perovskite Films for Perovskite Solar Cells
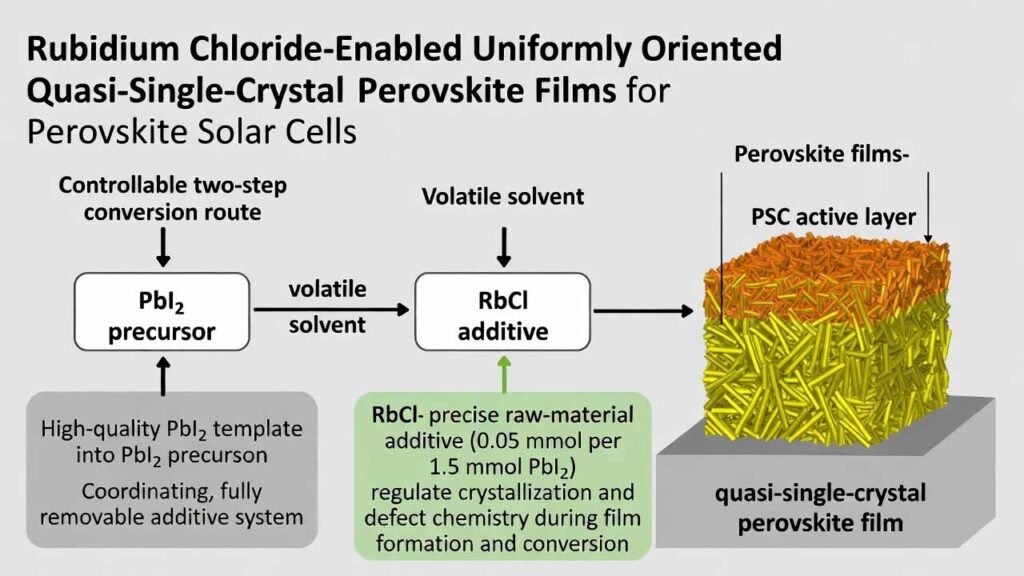
The key manufacturing idea is simple: (i) build a high-quality PbI2 template using a coordinating, fully removable (volatile) additive system, and (ii) use RbCl as a precise raw-material additive (typical level: 0.05 mmol per 1.5 mmol PbI2) to further regulate crystallization and defect chemistry during film formation and conversion.
Rubidium Chloride (RbCl) Charge-Enhanced Induced-Junction (MIS/IL) Solar Cells for Low-Temperature Silicon Photovoltaics
This cell structure uses a p-type semiconductor substrate with a backside metal electrode and a front-side “induction layer” stack: transition layer (SiOx or SiOxNy), a charge-enhancement layer (e.g., RbCl), and an anti-reflection layer (SiNx). With an optimized charge-enhancement layer, the fixed positive charge density in the induction layer can reach the 1012–1013 cm-2 range while maintaining low interface-state density, improving surface passivation and device performance.
