Rubidium Chloride (RbCl) in Air-Processed 3D Perovskite Blue-Emissive Thin Films for PeLED Emission Layers
1) Overview
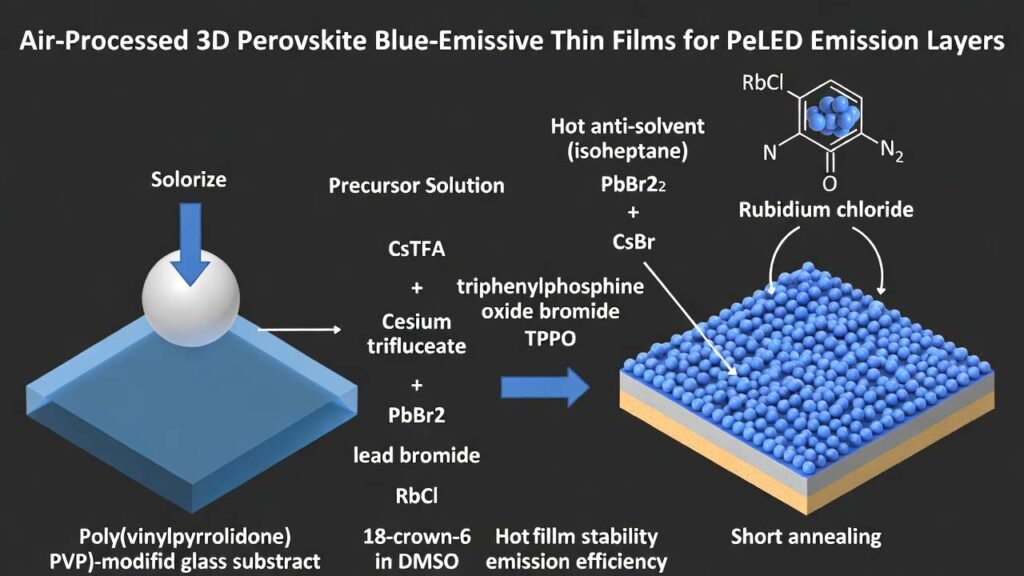
Metal-halide perovskites are attractive emissive-layer materials due to solution processability and tunable bandgap. A key barrier to scale-up is that many high-performance perovskite light-emitting films are prepared in inert environments (e.g., gloveboxes) to avoid moisture-driven defects during crystallization. The workflow below enables 3D perovskite blue-emissive thin films fabricated directly in air by combining: (i) a poly(vinylpyrrolidone) (PVP)-modified glass substrate, (ii) a DMSO-based heated precursor solution containing cesium trifluoroacetate (CsTFA), lead bromide (PbBr₂), rubidium chloride (RbCl), and 18-crown-6 (optionally with triphenylphosphine oxide (TPPO) and cesium bromide (CsBr)), and (iii) a hot anti-solvent (isoheptane) drip during spin-coating plus a short anneal.
By adjusting the CsTFA/CsBr ratio (while keeping PbBr₂ and RbCl fixed), a series of sky-blue perovskite films can be obtained, suitable as emissive layers for high-brightness blue PeLED development.
Safety note for R&D labs: Pb salts and aprotic solvents require appropriate PPE, ventilation, and compliant waste handling.
2) Detailed Experimental Procedure
- Glass substrates
- PVP (poly(vinylpyrrolidone))
- DMF (for PVP solution), DMSO (for perovskite precursor)
- CsTFA, PbBr₂, RbCl (rubidium chloride), 18-crown-6
- Optional additives: TPPO, CsBr
- Anti-solvent: isoheptane
- Cleaning: detergent/cleaning solution, ultrapure water, ethanol
- Ultrasonic bath, N₂ gun, UV-ozone system, O₂ plasma system
- Spin coater, hotplates (60–120 °C and 80–90 °C)
- Syringe + micropipette, 0.22 µm filter (chemical-resistant)
- PL setup (e.g., 365 nm excitation) for film screening
- Wipe the glass with a lint-free cloth wetted with detergent.
- Ultrasonicate sequentially for 15 min each in: (i) glass cleaning solution (detergent:DI water = 1:30), (ii) water, (iii) ethanol.
- Dry the substrate (e.g., bake dry) and blow with N₂.
- Treat the surface using UV-ozone (then reserve for coating).
- Apply O₂ plasma treatment for 600 s to activate the surface.
- Prepare a 4 mg/mL PVP solution in DMF (fully dissolve).
- Spin-coat PVP/DMF on the pre-treated glass at 5000 rpm for 30 s.
- Anneal at 120 °C for 5 min to form a uniform PVP interlayer.
Practical aim: the PVP interlayer improves wetting and supports more controlled nucleation/crystallization of the blue-emissive perovskite film in air.
Prepare the precursor in 1.0 mL DMSO, heat and stir at 60 °C for 4 h, then filter and store for use.
| Precursor set | Composition in 1 mL DMSO | Purpose of this tuning |
|---|---|---|
| Set 1 (baseline) |
CsTFA 0.1 mmol PbBr₂ 0.1 mmol RbCl 0.1 mmol 18-crown-6 5 mg |
Baseline 3D mixed-halide blue film formulation using RbCl as a key alkali-halide component. |
| Set 2 (additive passivation) |
CsTFA 0.1 mmol PbBr₂ 0.1 mmol RbCl 0.1 mmol 18-crown-6 11 mg TPPO 30 mg |
Increase additive-driven defect control while retaining the same RbCl loading. |
| Set 3–5 (blue wavelength tuning) |
PbBr₂ 0.1 mmol RbCl 0.1 mmol 18-crown-6 11 mg TPPO 30 mg Set 3: CsTFA 0.07 mmol + CsBr 0.03 mmol Set 4: CsTFA 0.05 mmol + CsBr 0.05 mmol Set 5: CsTFA 0.03 mmol + CsBr 0.07 mmol |
Adjust Br-related composition via CsTFA/CsBr ratio to shift blue emission while keeping RbCl constant for reproducibility. |
- Add the weighed salts/additives into 1.0 mL DMSO.
- Heat and stir at 60 °C for 4 h until fully dissolved.
- Filter the solution (fine filter) to remove particulates.
- Store the filtered precursor for coating (kept protected from moisture; an inert container/headspace is recommended).
- Preheat the precursor solution and isoheptane anti-solvent at 90 °C for ≥5 min.
- In ambient air, dispense the heated precursor onto the PVP-modified substrate.
- Spin-coat at 4000 rpm for 90 s.
- At 35 s remaining during spin, drip 150 µL of heated isoheptane onto the spinning substrate.
- Anneal at 80 °C for 1 min to form the 3D perovskite blue-emissive film.
The combination of heated precursor and heated anti-solvent is designed to accelerate film formation and reduce moisture interference during crystallization in air.
- Measure PL under 365 nm excitation.
- Representative sky-blue emission peaks reported for this workflow fall around ~478–483 nm with narrow FWHM values (~15–18 nm).
- Example PLQY outcomes vary by formulation; one additive-optimized set reported higher PLQY than the baseline set.
Use these values as process targets for troubleshooting (film uniformity, pinholes, phase separation, moisture sensitivity), not as guaranteed specifications.
3) Comparison vs Traditional Production Routes (Summary)
- Film formation inside inert atmosphere (N₂/Ar glovebox) to avoid water/oxygen impact.
- Higher capital and operating complexity for scaling (glovebox throughput limits, maintenance, moisture control).
- In air, the same chemistries often suffer from non-uniform nucleation, higher defect density, and reduced PL efficiency.
- PVP interlayer promotes controlled wetting and crystallization on glass, improving film-forming tolerance in air.
- Heated precursor + heated isoheptane anti-solvent accelerates nucleation/solidification, helping suppress water-vapor disruption during the critical crystallization window.
- Additive set (18-crown-6, TPPO) + halide/cesium-source tuning (CsTFA/CsBr) targets defect reduction and emission tuning while preserving a 3D perovskite framework.
- Production relevance: simpler ambient processing can align better with batch coating and future large-area coating concepts compared with fully inert-only routes.
4) Why Rubidium Chloride (RbCl) Is a Key Raw Material Here
In this formulation, RbCl is not a minor “optional” salt—it is a defined precursor component (kept constant at 0.1 mmol in multiple formulations) that supports repeatable blue-film formation in air processing.
What RbCl contributes in practice- Rb⁺ incorporation control: Rubidium cations can act as an alkali component within the precursor chemistry that supports more stable, reproducible crystallization behavior for 3D perovskite blue-emissive films.
- Halide environment support: Chloride is introduced directly via RbCl, participating in the mixed-halide precursor environment that helps push emission toward the blue region when paired with bromide-containing lead salt and cesium-source tuning.
- Synergy with 18-crown-6 and TPPO: 18-crown-6 can coordinate alkali cations and help regulate ionic activity during nucleation; TPPO is commonly used to interact with under-coordinated sites. With RbCl present at a fixed dose, these additives can work more consistently across batches.
- Manufacturing reproducibility lever: Because the recipe fixes RbCl while adjusting CsTFA/CsBr, RbCl becomes a “constant anchor” that reduces the number of moving variables when tuning blue wavelength and PL performance.
- High purity (commonly 99.9% grade for optoelectronic R&D), with controlled alkali/metal impurities.
- Low moisture and good storage/packaging (moisture pickup can destabilize DMSO precursor consistency and air coating repeatability).
- Low Na/K contamination if you are benchmarking ionic additives precisely.
- Good lot-to-lot consistency (assay, insolubles) to keep emission tuning focused on CsTFA/CsBr rather than raw material drift.
If your goal is air fabrication of blue perovskite emission layers, treating rubidium chloride (RbCl) as a controlled, high-spec raw material (not a commodity salt) is one of the simplest ways to improve reproducibility when scaling from small coupons to larger substrates. The synthesis method mentioned in this article references patent document number CN202510092319.X
