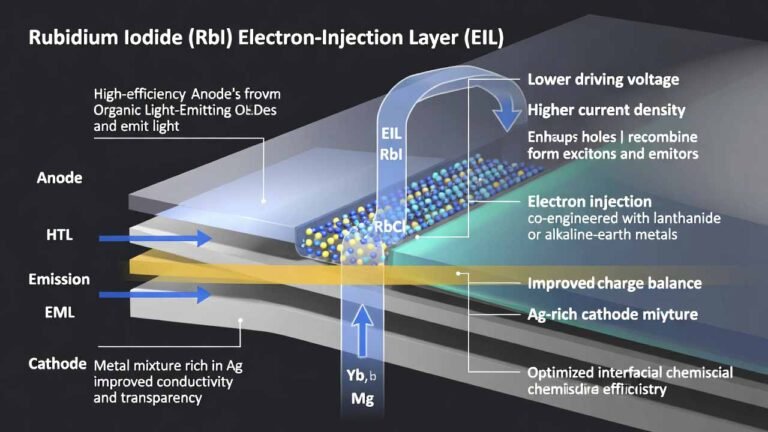
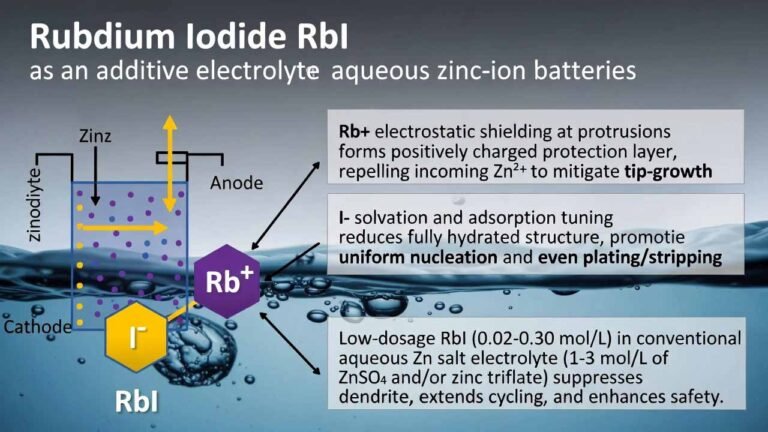
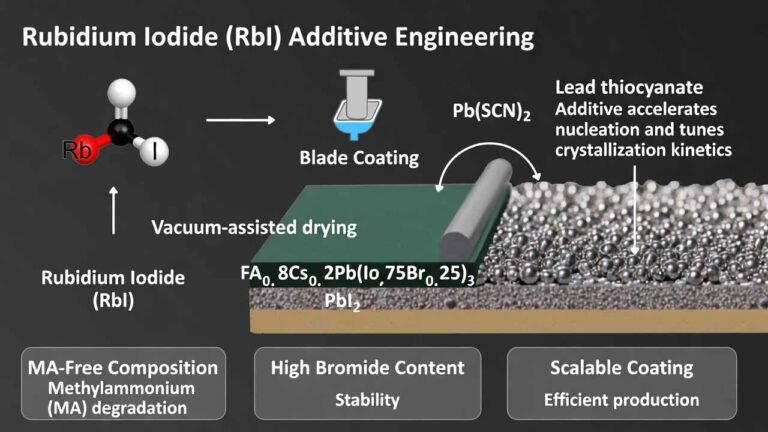
Rubidium iodide (RbI)
CAS Number: 7790-29-6 , EC Number:232-198-1
Brand Name : DQ
Percentage assay:99.5%
Package Information: Bottle or 25kg/drum or customized packing
Rubidium iodide Price
Product Information
| Parameter | Value or Description | Units or Conditions | Remarks |
|---|---|---|---|
| Molecular Formula | RbI | N/A | Rubidium Iodide |
| Molecular Weight | 212.37 | g/mol | |
| Appearance | White, crystalline solid | N/A | |
| Melting Point | 642 °C | Celsius | |
| Boiling Point | 1300 °C | Celsius | |
| Density | 3.35 g/cm³ | g/cm³ | At 25°C |
| Solubility | Highly soluble in water | N/A | |
| Refractive Index | 1.784 | N/A | |
| Electrical Conductivity | High | S/m | |
| Thermal Conductivity | 7.2 W/(m·K) | W/(m·K) | At 25°C |
| Specific Heat Capacity | 0.51 J/(g·K) | J/(g·K) | |
| Chemical Stability | Stable under normal conditions | N/A | |
| Common Uses | Photographic emulsions, scintillation counters, etc. | N/A |
Rubidium iodide Formul
| Reaction Type | Reactants | Products | Balanced Equation |
|---|---|---|---|
| Formation | Rubidium (Rb) + Iodine (I₂) | Rubidium Iodide (RbI) | 2Rb+I2→2RbI |
| Dissolution in Water | Rubidium Iodide (RbI) + Water (H₂O) | Rb⁺ + I⁻ ions in solution | RbI→Rb++I− |
| Reaction with Acid | Rubidium Iodide (RbI) + Hydrochloric Acid (HCl) | Rubidium Chloride (RbCl) + Hydroiodic Acid (HI) | RbI+HCl→RbCl+HI |
| Double Displacement | Rubidium Iodide (RbI) + Sodium Chloride (NaCl) | Rubidium Chloride (RbCl) + Sodium Iodide (NaI) | RbI+NaCl→RbCl+NaI |
Applications of Rubidium iodide
| Application Area | Specific Use | Industry | Additional Notes |
|---|---|---|---|
| Scientific Research | Used in spectroscopy and various lab experiments | Academia | Often used in small quantities |
| Electronics | Used in photodetectors and other optoelectronic devices | Technology | |
| Medicine | Potential use in radiopharmaceuticals | Healthcare | Still under research |
| Manufacturing | Used as a starting material for the synthesis of other rubidium compounds | Chemical Industry |
Safety Information
| Safety Category | Description |
|---|---|
| Physical Hazards | Non-flammable, but may react with other substances. |
| Health Hazards | May cause skin and eye irritation. Ingestion can lead to gastrointestinal issues. |
| Environmental Hazards | May be harmful to aquatic life. |
| Personal Protection | Use gloves, safety goggles, and lab coat. Ensure adequate ventilation. |
| Storage | Store in a cool, dry place away from incompatible materials like strong acids. |
| Disposal | Dispose of in accordance with local, state, and federal regulations. |