Introduction to Rubidium
Rubidium was discovered in 1861 by German chemists Robert Bunsen and Gustav Kirchhoff. It was identified through flame spectroscopy, a technique that was relatively new at the time. Rubidium plays a vital role in various applications, from industrial processes to scientific research.
This device is a compact, opto-electronic coherent population trapping (CPT) magnetometer built around a sealed rubidium (Rb) atomic vapor cell. A dual-frequency coherent laser field is converted into circularly polarized light and sent through an Rb vapor interaction region; the transmitted (or reflected) optical power is detected by an integrated photodiode. When the two optical frequencies sati…
This configuration generates a narrow, directional 420 nm blue laser from rubidium (Rb) vapor by two-photon pumping at 778 nm inside a sealed vapor cell. A hollow-core photonic crystal fiber (HCPCF) is mounted inside the Rb cell and acts as an in-cell resonant/feedback and long-interaction structure: it extends the effective gain length and repeatedly guides the interacting wavelengths, helping th…
Low-dimensional magnets are attractive for next-generation information technologies because magnetic order can persist in extremely thin geometries, enabling dense data storage concepts, device miniaturization, and new quantum/spintronic functionalities. A major bottleneck is that many established low-dimensional magnets are inorganic and/or rely on complex fabrication routes (e.g., exfoliation, e…
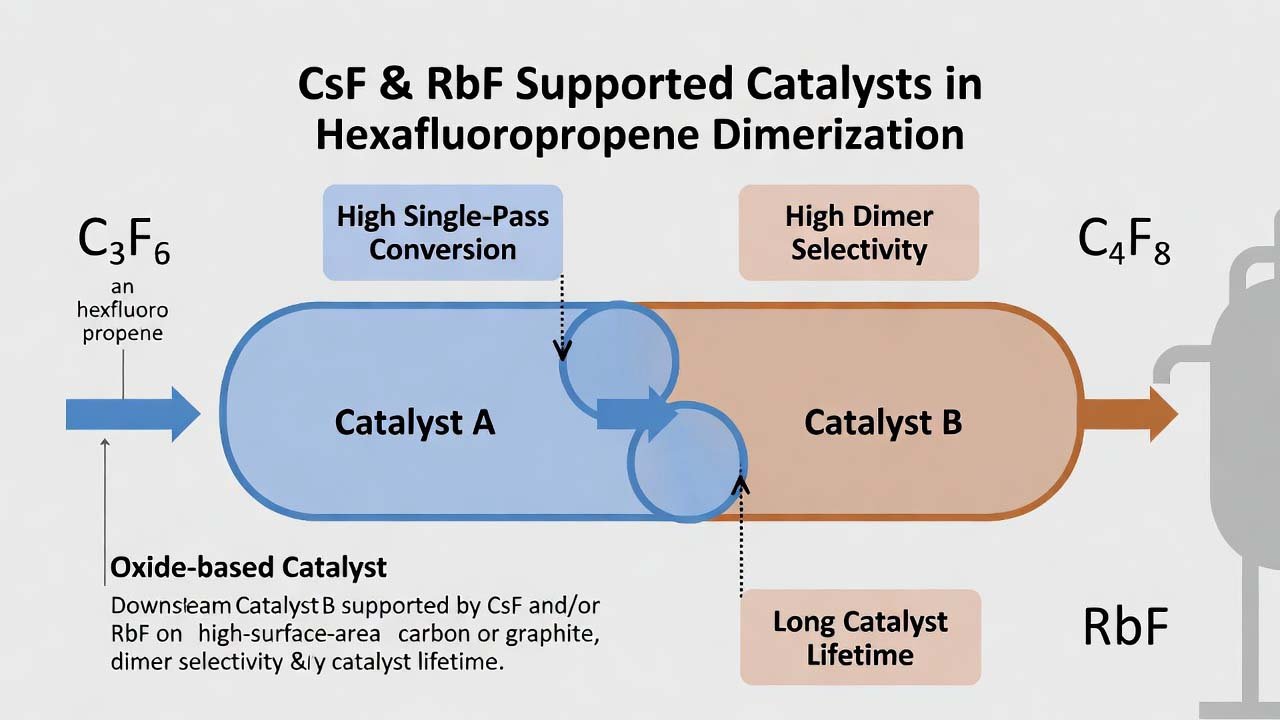
Hexafluoropropene dimer (HFPD) is a key fluorochemical intermediate because the remaining C=C bond enables downstream synthesis of multiple fluorinated derivatives used in pharmaceuticals, fluorosurfactants, specialty solvents, and replacement agents in fire protection chemistries. The practical challenge in HFP dimerization is achieving high single-pass conversion, high dimer selectivity (suppres…
Flux-coated Zn–Al brazing rings are widely used for joining copper–aluminum tubing in refrigeration systems (e.g., compressors, condensers, and capillary lines), where stable wetting, high fillability, and consistent joint strength are required. This manufacturing approach builds a three-layer ring: an inner pure Zn tube that defines a small inner diameter, a Zn–Al alloy core layer (target alloy: …
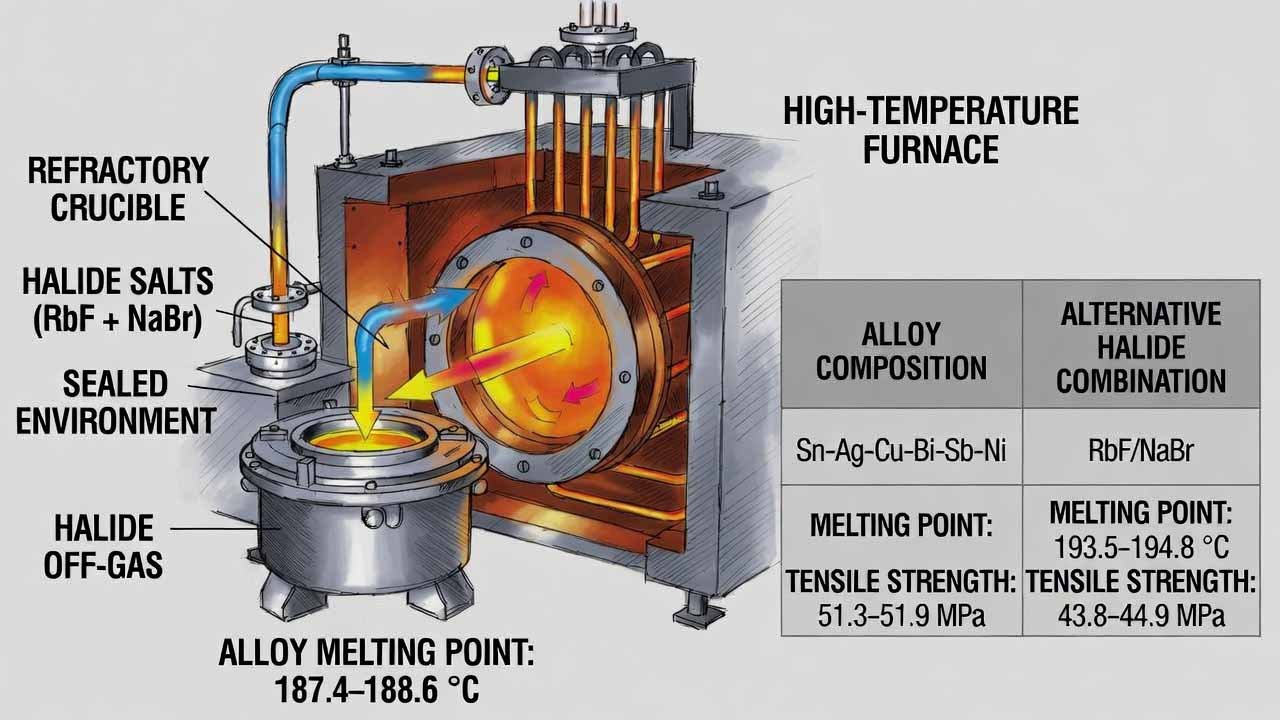
This workflow produces a lead-free tin-based multi-element solder alloy (Sn-Ag-Cu-Bi-Sb-Ni) designed for stable melting behavior and improved joint reliability. The core technical lever is introducing rubidium fluoride (RbF) together with sodium bromide (NaBr) during a sealed, high-temperature melt. Under sealed conditions, the halide additives generate a reactive halide-rich atmosphere that impro…
Aluminum and aluminum-alloy brazing in ambient atmospheres is fundamentally limited by the rapid formation of stable surface oxides. A high-activity fluoride-based flux is therefore essential to disrupt oxides, promote wetting, and enable reliable joints. Compared with conventional Nocolok-type fluxes (K3AlF6–KAlF4 / KAlF4) that typically operate near ~560–580 °C and often depend on controlled atm…
Crystalline silicon (c-Si) remains the most mature and widely deployed photovoltaic platform. The next efficiency gains increasingly depend on reducing carrier recombination and contact resistance at the c-Si/metal interface, while simplifying process complexity and lowering cost. Conventional architectures (e.g., direct metal contacts on lightly doped silicon in standard PERC) can suffer from int…