Introduction to Rubidium
Rubidium was discovered in 1861 by German chemists Robert Bunsen and Gustav Kirchhoff. It was identified through flame spectroscopy, a technique that was relatively new at the time. Rubidium plays a vital role in various applications, from industrial processes to scientific research.
A practical way to restore stability is to add a soluble alkaline agent that is compatible with the sulfuric-acid vanadium electrolyte system and does not form precipitates. Rubidium hydroxide (RbOH) is one option in this alkaline set; it supplies strong, fast neutralization via OH− while keeping added species fully soluble in sulfate media (rubidium salts remain soluble under typical VRFB acid ra…
The catalyst is built from three functional parts: (i) a high-surface-area, hydrophobic and selective carrier (ZSM-5 + TiO2 + modified diatomite, further treated with a silane coupling agent), (ii) an active phase generated from copper/nickel/chromium nitrates synergized with rubidium nitrate through mixing, short combustion, and calcination to create highly dispersed mixed oxide catalytic centers…
This process performs ion exchange by immersing the glass in an aqueous ion-exchange solution under a microwave hydrothermal environment. The solution is formulated from potassium nitrate, potassium silicate, rubidium nitrate, and cesium nitrate. Microwave hydrothermal conditions accelerate ion transport and diffusion, improving the exchange rate and reducing overall processing time while maintain…
This workflow integrates rubidium nitrate (RbNO3) as the active salt on activated carbon and uses dynamic oxygen dosing to continuously balance two competing needs: (i) oxidizing deposited carbon to keep pores open and active sites exposed, and (ii) preventing excessive oxidation (burn-off) of the activated carbon support.
In this workflow, rubidium nitrate (RbNO3) is a key feedstock because it is simultaneously: (1) the only explicitly defined nitrate (NO3−) source for building the mixed-anion framework, and (2) a rubidium (Rb+) contributor that helps lock stoichiometry and phase formation when paired with boron–oxygen precursors. For R&D and process scale-up, nitrate incorporation control is often the difference b…
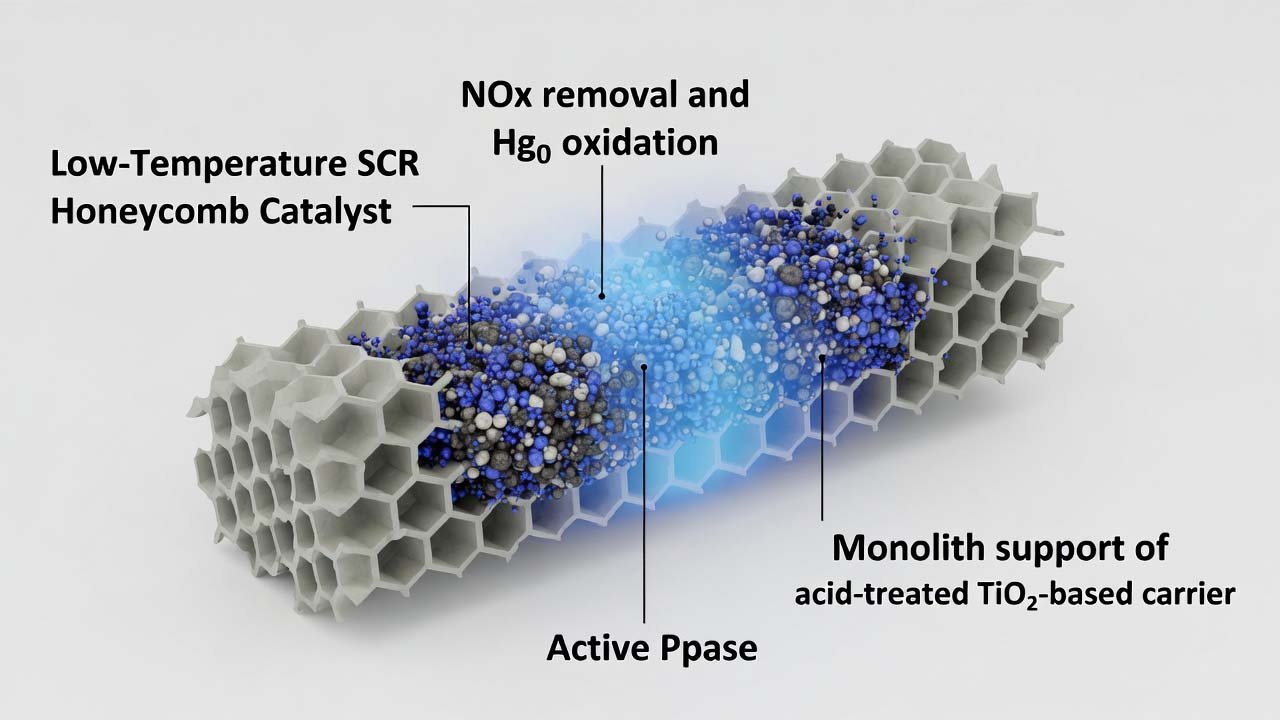
This process prepares a modified honeycomb SCR catalyst composed of a porous monolith support (acid-treated TiO2-based carrier) and a multi-component active phase introduced by impregnation. Rubidium nitrate (RbNO3) is incorporated directly into the carrier formulation to enhance sulfur resistance and stability under low-temperature conditions, while V–W–Mo and Mn/Ce plus trace noble-metal nitrate…
A practical solution is to combine ZnCl₂ (as a microwave absorber) with a promoter to amplify microwave heating and raise the effective pyrolysis temperature rapidly. In this workflow, Rubidium Carbonate (Rb₂CO₃) can be selected as the promoter to enhance the heating efficiency of the ZnCl₂-assisted system, enabling short, air-atmosphere microwave treatment to produce highly carbonized carbon-supp…
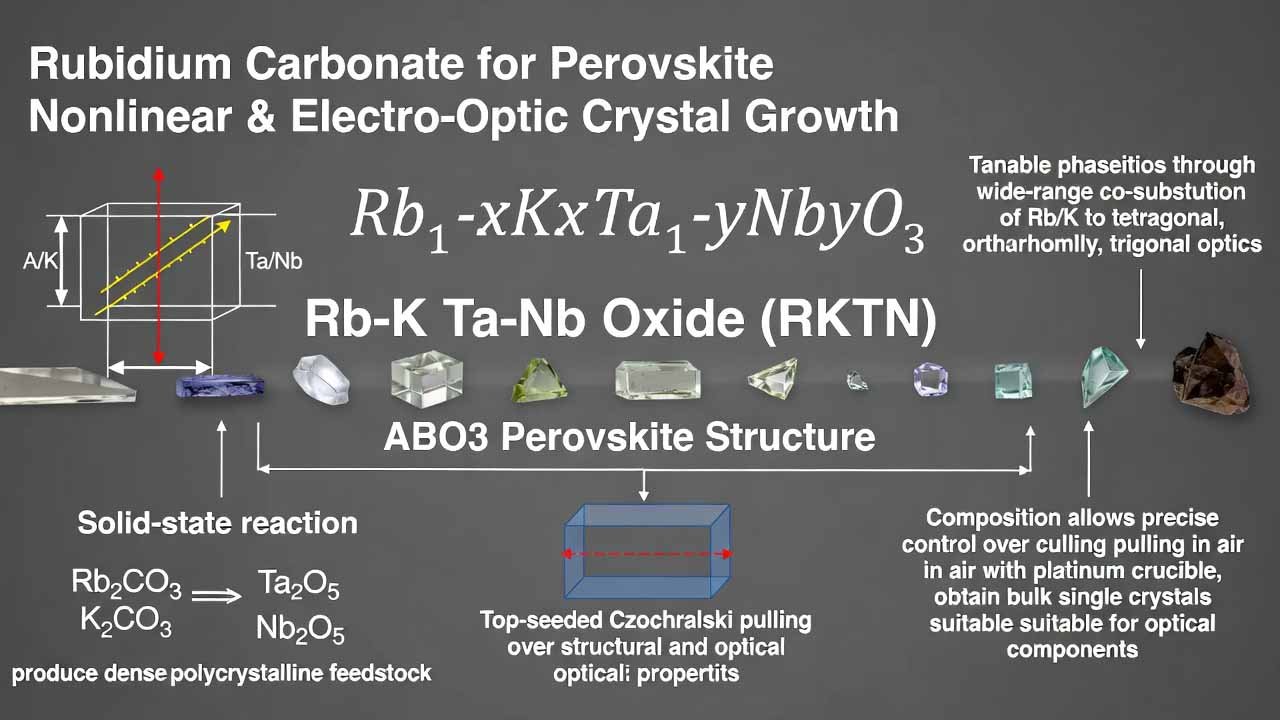
A practical manufacturing route combines: (i) solid-state reaction using Rb2CO3, K2CO3, Ta2O5, Nb2O5 to produce dense polycrystalline feedstock, and (ii) top-seeded Czochralski pulling in air (platinum crucible) to obtain bulk single crystals suitable for optical component fabrication.