Introduction to Rubidium
Rubidium was discovered in 1861 by German chemists Robert Bunsen and Gustav Kirchhoff. It was identified through flame spectroscopy, a technique that was relatively new at the time. Rubidium plays a vital role in various applications, from industrial processes to scientific research.
Rb2B3O3F4(OH) is a rubidium borate-fluoride-hydroxyl compound that can be grown into a stable nonlinear optical (NLO) single crystal for ultraviolet frequency conversion. The crystal belongs to an orthorhombic system (space group Ama2) and exhibits a deep-UV cutoff edge below 200 nm, making it attractive for short-wavelength UV generation in all-solid-state laser platforms.
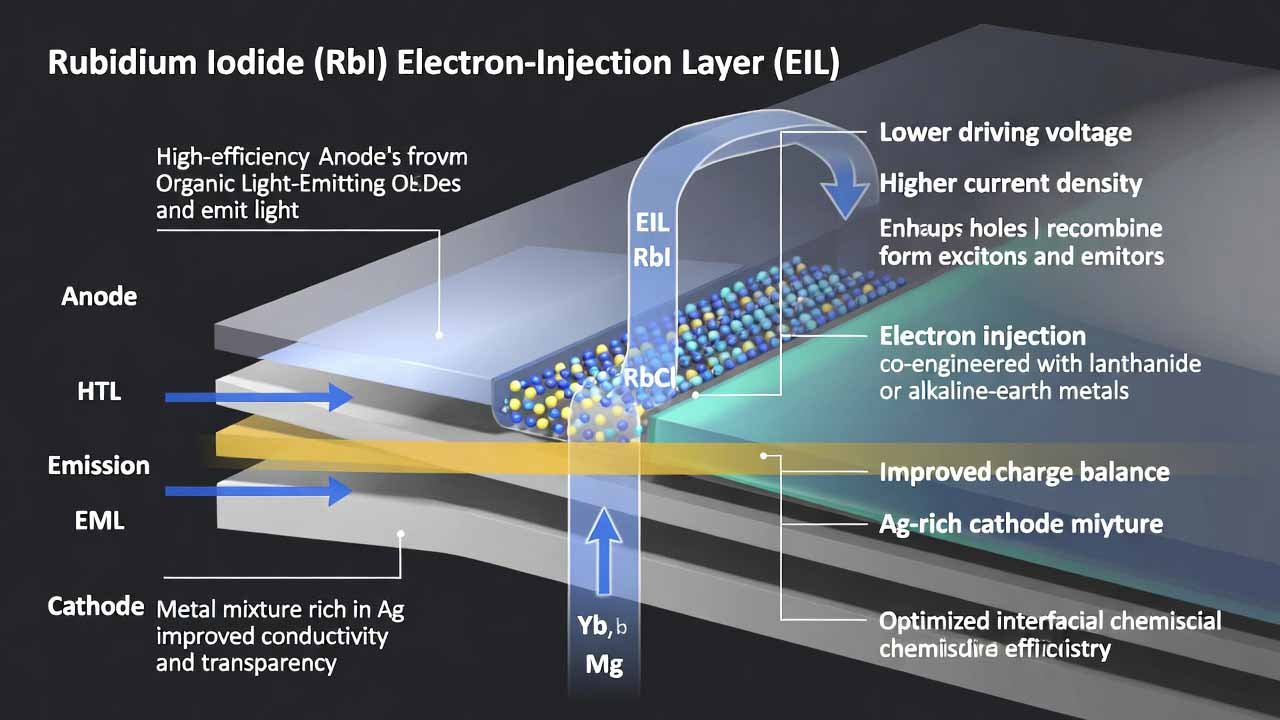
Organic light-emitting devices (OLEDs) are self-emissive stacks where holes (from the anode) and electrons (from the cathode) recombine in the emission layer (EML) to generate excitons and light. Beyond the EML, functional interlayers—hole injection/transport, electron transport, and especially the electron injection layer (EIL)—are decisive for lowering driving voltage, improving charge balance, …
This electrolyte strategy introduces rubidium iodide (RbI) as a low-dosage additive (0.02–0.30 mol/L) into a conventional aqueous Zn salt electrolyte (1–3 mol/L of ZnSO4 and/or zinc triflate). RbI delivers a dual-ion interface regulation effect
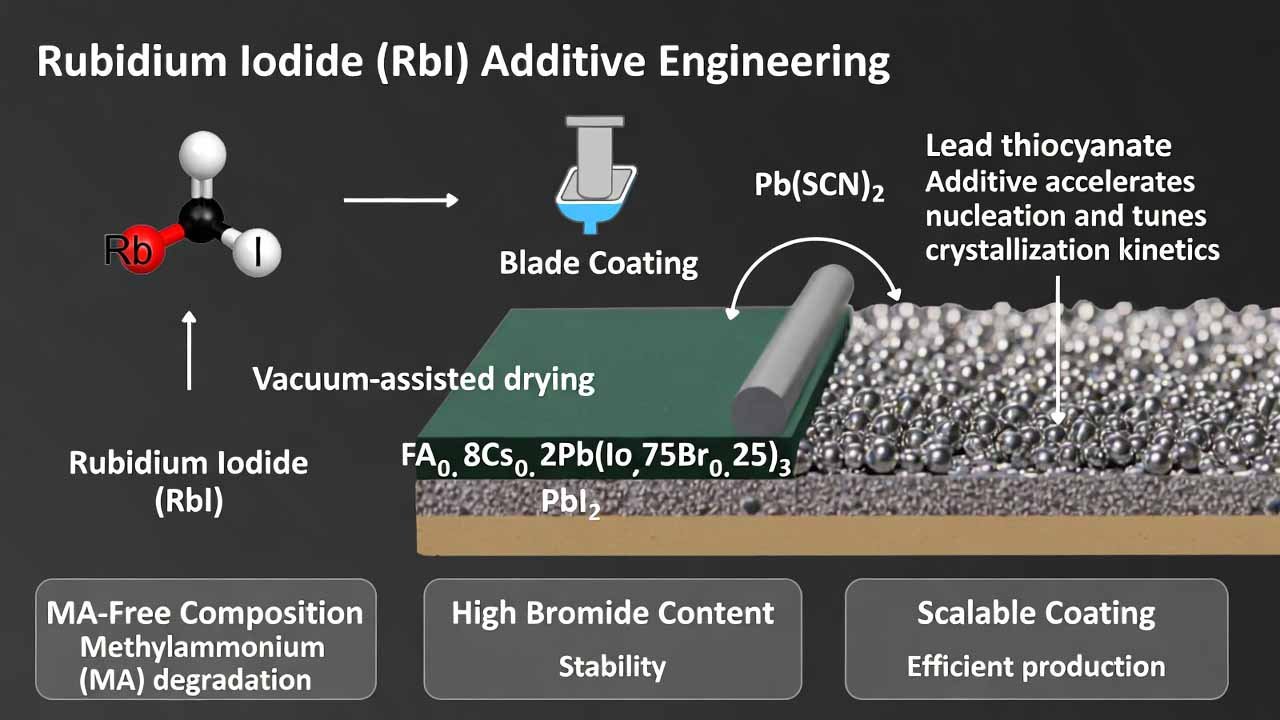
Perovskite solar cells (PSCs) are attractive for next-generation photovoltaics because they can be made by solution processing, potentially lowering capex and enabling large-area manufacturing. A core scale-up bottleneck is forming uniform, dense perovskite films over larger substrates without sacrificing device performance or stability—especially when moving from small-area lab spin coating to sc…
The approach here uses a methylammonium (MA)-free wide-bandgap composition FA0.8Cs0.2Pb(I0.75Br0.25)3 (bandgap ≈ 1.7 eV) to avoid MA-related degradation risk, and applies an additive pair: Pb(SCN)2 plus a rubidium salt (preferably rubidium iodide, RbI). Pb(SCN)2 accelerates nucleation and tunes crystallization kinetics, while the rubidium salt works synergistically to improve grain coalescence, su…
This process builds a dark-pipe-specific antibacterial additive by first using rubidium hydroxide (RbOH) aqueous solution to dissolve lignin and form a rubidium–lignin salt (rubidium ligninate) with highly uniform rubidium distribution at the molecular level. A controlled oxygen-atmosphere thermal treatment then converts the precursor into a brown/dark powder that blends efficiently with carbon bl…
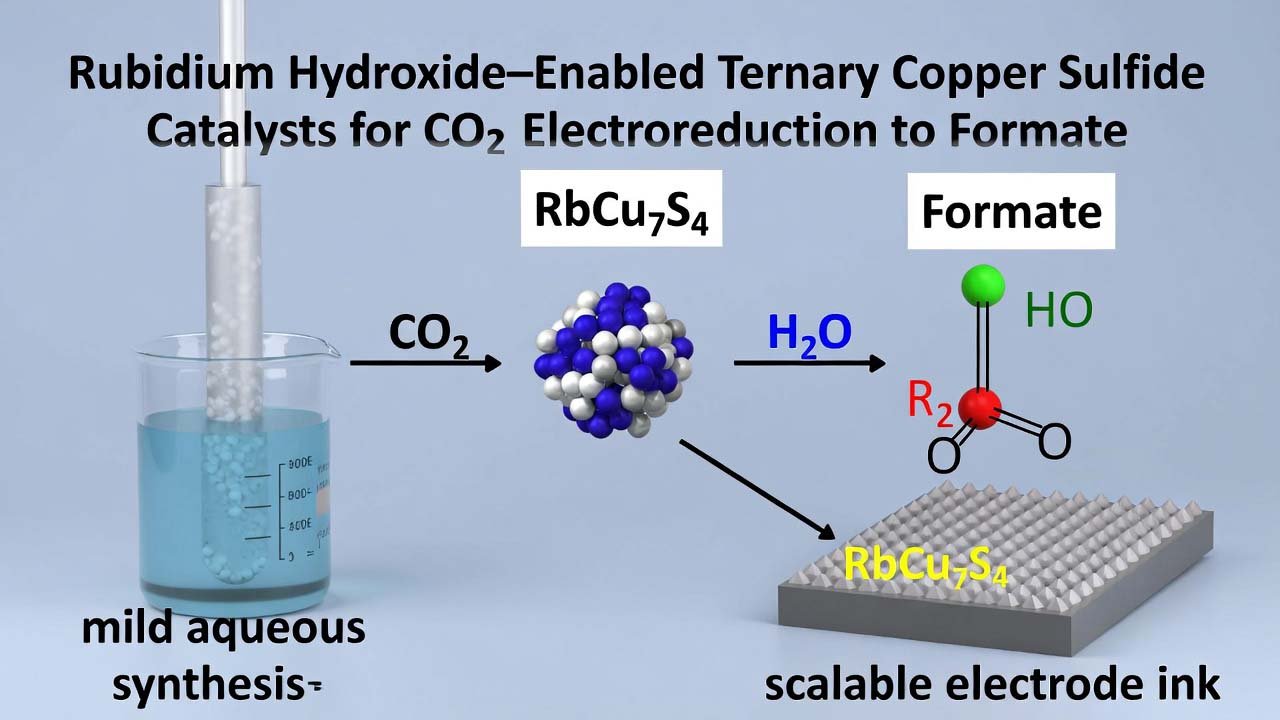
This workflow describes a low-temperature solution synthesis of ternary copper sulfide catalysts (M–Cu–S, M = Na, K, Rb), followed by electrode fabrication and CO2 electroreduction testing. The rubidium-containing phase (RbCu7S4) is produced by using rubidium hydroxide (RbOH) as the alkali metal source in water, enabling incorporation of Rb+ into the catalyst lattice. This helps the catalyst maint…
This workflow prepares rubidium metasilicate via an aqueous reaction using rubidium hydroxide (RbOH) as the rubidium source, followed by freeze-drying and inert-gas annealing to obtain a stable powder suitable for detector fabrication. The approach emphasizes process control (pH, clarity/transmittance, moisture, annealing atmosphere, and cooling rate) to improve product consistency and yield, and …